Home
This is a community of researchers who are interested in supporting the generation of more and better evidence to drive improvements in health across the globe. Clinical research needs evidence-led improved methods.
What is Methodology Research? Methodology Research is research about the way we design, conduct, analyse, report and interpret research studies. Conducting methodology research studies will generate evidence-led improvements in the way we design and run studies. Methodology Research is research on research.
Clinical trials methodology research and the Global Health Working Group of the MRC/NIHR Trials Methodology Research Partnership (TMRP)

Fully funded PhD opportunitiesAre you interested in medical research that matters to patients and the NHS? Are you interested in PhD projects in a wide array of disciplines, such as: Psychology, social science & qualitative methods, Statistics & mathematics, Informatics, data science & computer science, Health economics or Epidemiology & health services research? The Trials Methodology Research Partnership (TMRP) DTP is a collaborative PhD programme funded by MRC-NIHR. It focuses on enhancing how clinical trials are designed, conducted, analysed, and reported—ultimately aiming to improve patient care. It offers fully funded PhD opportunities in the disciplines previously mentioned. APPLICATIONS CLOSED JANUARY 2026 | START DATE: OCTOBER 2026 |

NEW COURSE
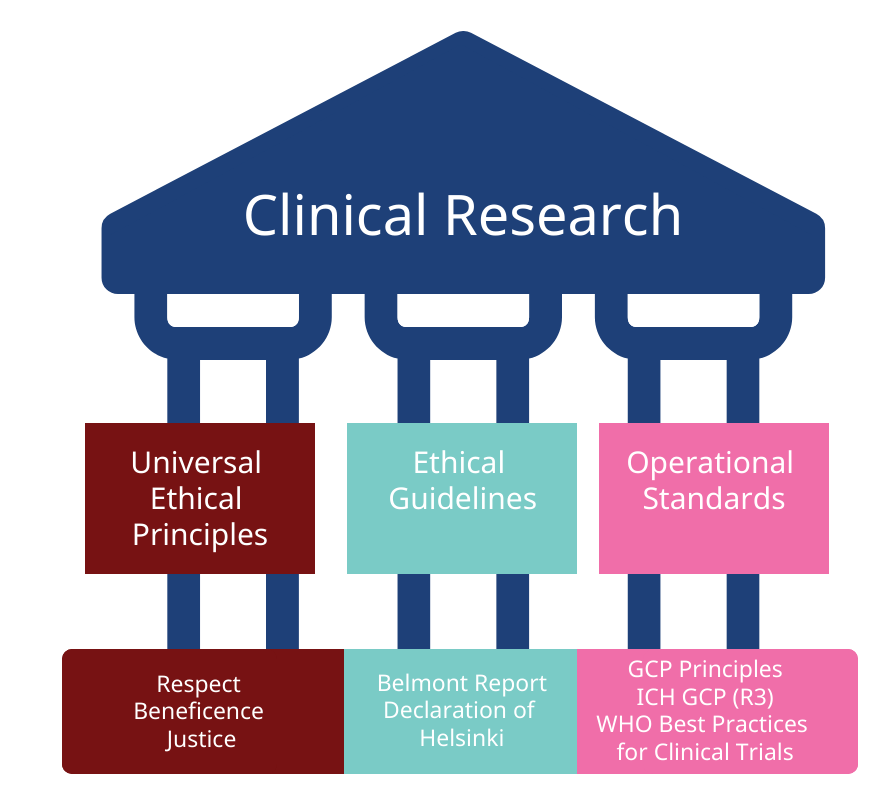
WHO Good Practices for Clinical Trial Design and Implementation
This course supports understanding and application of key ethical, scientific principles for designing and conducting high-quality, impactful clinical trials.
|
|